 Her interests include ultrasound, powerlifting, cooking, relaxing, reading nonfiction, enjoying the outdoors, staring at the stars, and lingering on the coast. In July of 2020, she began her Ultrasound Fellowship at USF. She completed medical school at Campbell University College of Osteopathic Medicine and did her residency training at Hackensack University Medical Center in New Jersey. Hansen was born in New Jersey but grew up in Clearwater, FL. Effect of VTILVOT variation rate on the assessment of fluid responsiveness in septic shock patients. Wang J, Zhou D, Gao Y, Wu Z, Wang X, Lv C. Rationale for using the velocity-time integral and the minute distance for assessing the stroke volume and cardiac output in point-of-care settings. Measurement site of inferior vena cava diameter affects the accuracy with which fluid responsiveness can be predicted in spontaneously breathing patients: a post hoc analysis of two prospective cohorts. Randomized Controlled Trial of Ultrasound-guided Fluid Resuscitation of Sepsis-Induced Hypoperfusion and Septic Shock. Musikatavorn K, Plitawanon P, Lumlertgul S, et al. Does Respiratory Variation in Inferior Vena Cava Diameter Predict Fluid Responsiveness: A Systematic Review and Meta-Analysis. Long E, Oakley E, Duke T, Babl FE Paediatric Research in Emergency Departments International Collaborative (PREDICT). If VTI is low (10-15%, your patient is likely a fluid responder.ġ. I would trace multiple waveforms and average them for accuracy.Ĥ. Under your advanced cardiac measurements, you should have a VTI option to trace your waveform like in the image below. Press your PWD button again and you should obtain a nice negative waveform tracing (flow is away from your probe, and therefore it displays a negative tracing) as seen below.ģ. Ensure the flow through the LVOT is parallel to your US beam (the apex of heart should be at the top of your screen, see image below).Ģ. Start by obtaining an apical-5 chamber view and placing a pulsed wave doppler (PWD) gate right at the ventricle side of the LVOT (left ventricular outflow tract) approximately 1 cm from the valve. This will take some practice! Newer ultrasound machines have a cardiac calculations package as well as artificial intelligence (auto-VTI) that can measure VTI for you, making your job easier!ġ. So how can you start using cardiac VTI in your ED?įirst, work on getting the proper ECHO views and messing around with the spectral doppler on your US machine. If I’ve lost you in the equations, take a moment to review this great YouTube video! When VTI increases, you can infer that your stroke volume (LVOT area x VTI = SV) and cardiac output do as well. 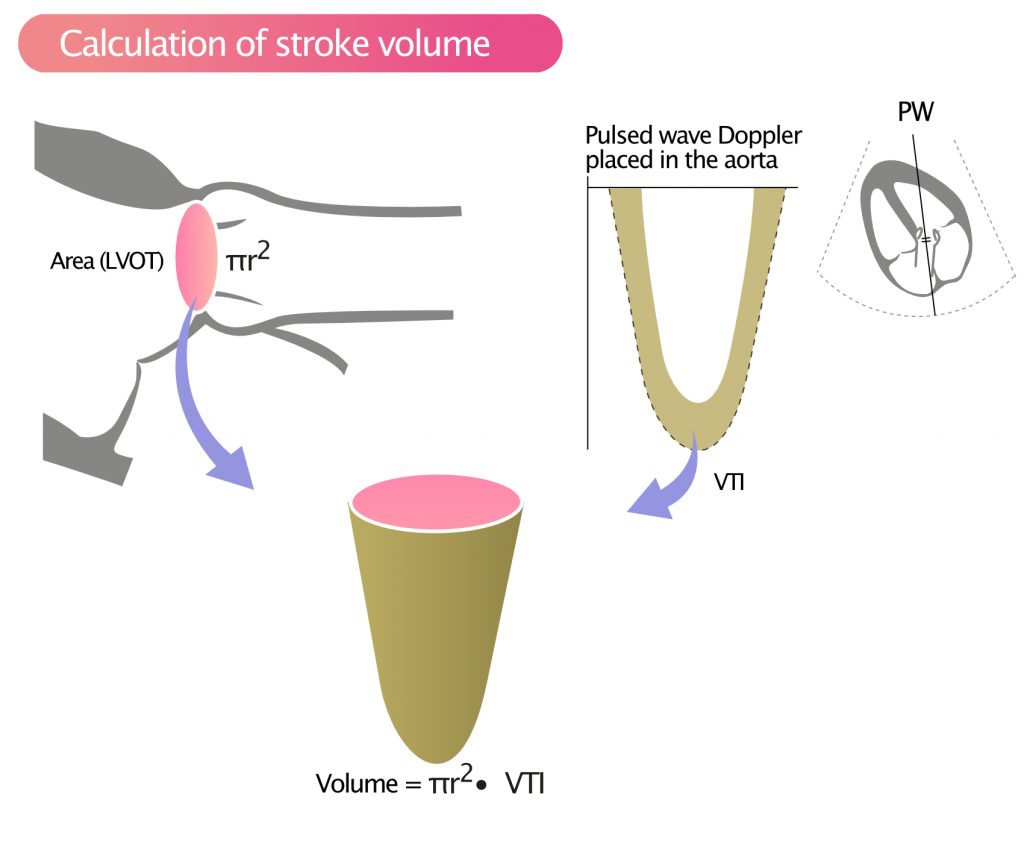
Basically, cardiac VTI is a component of the cardiac output equation as CO= LVOT area x VTI x HR. Why not jump straight to a derivative of CO?Įmerging evidence suggests cardiac velocity time integral (VTI), a measurement obtained by ultrasound, can be used to predict fluid responsiveness. Many studies use cardiac output as the gold standard to which they test against. It’s an added bonus if this can be done rapidly and through a non-invasive approach. Our goal is essentially to identify patients who will increase their cardiac output (CO) when given fluids. Unfortunately, studies demonstrated variable predictability and limited benefit. 
Assessing IVC respiratory variation became a popular approach for predicting fluid responsiveness. As clinicians shifted away from measuring IVC diameter, there was some thought that maybe respiratory variation of the IVC would be a better predictor than IVC diameter. A 2020 systematic review and meta-analysis in the Journal of Intensive Care Medicine by Ordo et al found a pooled sensitivity and spec of 0.71 and 0.75, respectively. Recently, there has been a trend away from using ultrasound interrogation of the IVC diameter to determine fluid responsiveness. Using the IVC for Fluid Responsiveness is Falling Out of Favor 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
